|
12/7/2023 0 Comments Troubleshooting western blotFor example, when assessing a blot, the band from one sample may appear twice as bright as another sample. It is essential, especially when trying to compare protein expression between different samples, to know how much sample has been loaded as this may not be apparent from the blot alone. It is also important to load appropriate control samples and size marker ladders to enable interpretation of the final blot. 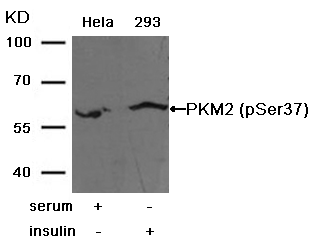
Solids will impair the running of the gel and it is likely your protein of interest will remain in the stacking gel. If your protein of interest is in the insoluble fraction (e.g., cell membrane-bound proteins) investigate pretreatment methods to liberate and solubilize it first. For a clean image, samples are centrifuged to remove any solids, in order to load only the soluble fraction. The specific separation method chosen will depend on the aim of the analysis. This is typically achieved by protein electrophoresis, such as sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS-PAGE) or native PAGE, which separates proteins based on their molecular weight or charge. Cell line/tissue with proven negative signal.Figure 1: Overview of a western blot protocol.īefore a western blot can be performed, the proteins in the sample must be separated.Samples targeted with RNA interference.Cell line/tissue with proven positive signal.Samples from cells overexpressing the target protein.While the omission of control samples from a Western blot is not a cause of non-specific bands, their inclusion can tell you why you may be seeing them on your membrane. Know your protein of interest, band sizes can shift due to glycosylation, phosphorylation, precursor maturation, etc.Presence of post-translational modifications Higher percentage Tris-Glycine gels (up to 15%) should be used for smaller proteins (<20 kDa) or use Tris-Tricine gels. (See page 12 for our SDS-PAGE gel recipes). Lower percentage Tris-Glycine gels should be used for larger proteins, or use Tris-Acetate-based gels and buffers. Use fresh, sonicated, and clarified tissue extracts.Īlways include protease inhibitors (and phosphatase inhibitors for the detection of phosphorylated targets).Ĭheck for the presence of known isoforms in the literature or at .Ĭhange the gel percentage to suit the target protein’s MW. Tissue extracts tend to produce more non-specific bands and degradation products. Use freshly prepared sample kept on ice up until the addition of sample buffer and immediate heating to 95☌ for 5–10 minutes. The following should also be considered: Target protein abundance is lower than the threshold of nonspecific bindingĮnrich low-abundance proteins by immunoprecipitation, fractionation, etc. The troubleshooting tips for high background (uniform distribution) can also be applied to scenarios where non-specific but distinct bands appear on the Western blot membrane. Check different types and dilution of the detection reagent. 
(Milk and casein are phosphoprotein-rich.)įilm exposure too long / Detection reagent too sensitive Use a pre-adsorbed secondary antibody with reduced crossreactivity to unwanted species.įor phosphorylated protein detection, do not use milk-based buffers such as non-fat milk or casein buffer. Perform a secondary antibody-only control experiment (omit the primary incubation step). Non-specific binding of secondary antibody Ensure membrane does not dry out during the immunoblotting process.Include blocking reagent and Tween 20 in the primary antibody dilution buffer. Increase blocking time and/or temperature. Increase the concentration of blocking reagent (e.g., from 5 to 7%). High Background (Uniform Distribution) Antibody concentration too high
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
